- Show Menu
- Contact Us
- FAQs
- Reader Service
- Survey Data
- Survey Winners
- Testimonials
- Upcoming Events
- Webinars
- White Papers
New & Noteworthy
April 2018
Oseltamivir Phosphate (Tamiflu) Liquid Dosage Form
Amneal Pharmaceuticals
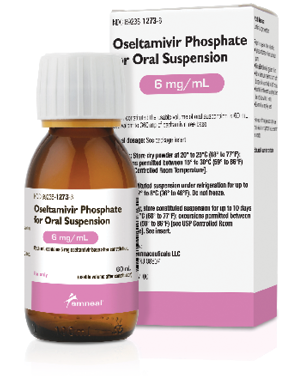
Amneal Pharmaceuticals launches oseltamivir phosphate for oral suspension, an AB-rated therapeutic equivalent for Tamiflu, in a 6 mg/mL strength. The powder-finished dosage form is made in the United States and packaged in bottles providing 60 mL of usable volume after constitution. Amneal’s oseltamivir phosphate for oral suspension joins its oral solid capsule form approved and launched in July 2017. The liquid dosage form is intended to help patients, particularly the elderly and young, who are unable to use an oral solid form.
Recent Popular Articles
About Us
Pharmacy Purchasing & Products Ridgewood Medical Media, LLC
Quick Links
Subscribe to Our Email Newsletter!
© 2005 - 2024 PP&P Magazine - Pharmacy Purchasing & Products.
All rights reserved.