- Show Menu
- Contact Us
- FAQs
- Reader Service
- Survey Data
- Survey Winners
- Testimonials
- Upcoming Events
- Webinars
- White Papers
New & Noteworthy
May 2018
Erythromycin Tablets
Amneal Pharmaceuticals
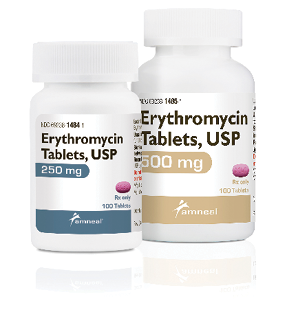
Amneal Pharmaceuticals has received FDA approval for erythromycin tablets USP, in 250 mg and 500 mg strengths. The product is a therapeutic equivalent for the reference listed drug (RLD) erythromycin tablets from Arbor Pharmaceuticals and is an immediate release oral tablet. They are packaged in 100-count bottles and are now available to wholesalers, distributors, and direct.
Recent Popular Articles
About Us
Pharmacy Purchasing & Products Ridgewood Medical Media, LLC
Quick Links
Subscribe to Our Email Newsletter!
© 2005 - 2024 PP&P Magazine - Pharmacy Purchasing & Products.
All rights reserved.