- Show Menu
- Contact Us
- FAQs
- Reader Service
- Survey Data
- Survey Winners
- Testimonials
- Upcoming Events
- Webinars
- White Papers
New & Noteworthy
March 2020
Emergency Gynecologic Methotrexate Kit
Edge Pharma, LLC
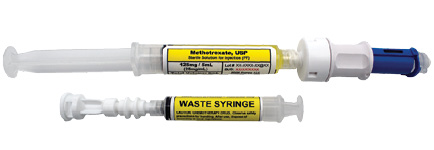
EdgePharma’s EmGynKit addresses the USP <800> compliance challenge affecting hospitals and clinics in preparing and administering patient-specific, body surface area-based doses of methotrexate up to 125 mg. The kit includes two syringes connected through a closed system drug-transfer device (CSTD). Body surface area based dosing is achieved by activating the CSTD, transferring the unneeded methotrexate into the waste syringe, deactivating the CSTD, and then detaching the waste syringe from the system.
Edge Pharma
Recent Popular Articles
About Us
Pharmacy Purchasing & Products Ridgewood Medical Media, LLC
Quick Links
Subscribe to Our Email Newsletter!
© 2005 - 2024 PP&P Magazine - Pharmacy Purchasing & Products.
All rights reserved.