- Show Menu
- Contact Us
- FAQs
- Reader Service
- Survey Data
- Survey Winners
- Testimonials
- Upcoming Events
- Webinars
- White Papers
New & Noteworthy
October 2010
Microbial Contamination Detection
Q.I. Medical, Inc
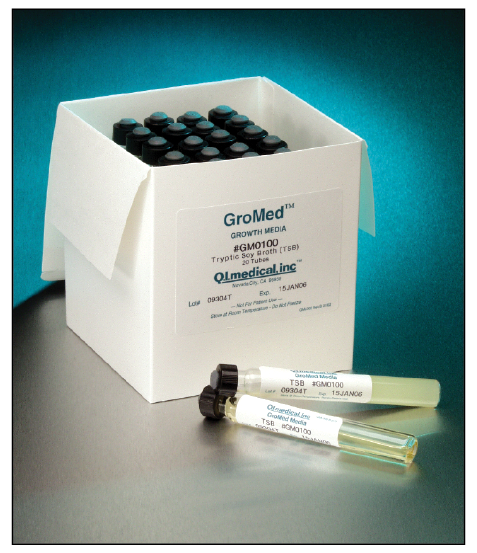
Q.I. Medical introduces GroMed, liquid media for aseptic technique testing and detecting microbial contamination in CSPs. Each glass test tube features both a screw cap and needle access port, and contains 10 mL of TSB growth media. Viscous sterile products can be introduced into the media via the screw cap. The injection port allows transfer of CSPs with enhanced aseptic technique. Designed to test CSPs from batches of less than 25 doses, all GroMed lots have certificates of analysis per USP standards.
Recent Popular Articles
About Us
Pharmacy Purchasing & Products Ridgewood Medical Media, LLC
Quick Links
Subscribe to Our Email Newsletter!
© 2005 - 2026 PP&P Magazine - Pharmacy Purchasing & Products.
All rights reserved.










