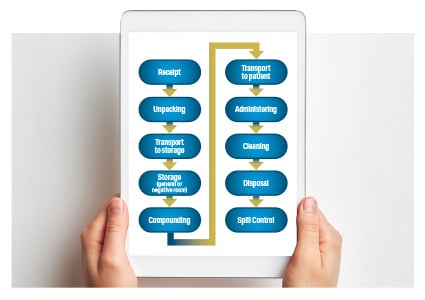
- Show Menu
- Contact Us
- FAQs
- Reader Service
- Survey Data
- Survey Winners
- Testimonials
- Upcoming Events
- Webinars
- White Papers
New & Noteworthy
November 2022
Propofol RFID Incorporation
Kit Check announces that its radio frequency identification (RFID) technology will be incorporated in the ready-to-administer propofol syringes offered by Genixus, a 503B outsourcing provider focused on providing acute and critical care medicines. The RFID-tagged propofol is available in 10 mL and 20 mL doses. Medications labeled with RFID tags at the manufacturer level provide item-level visibility of drug products throughout their dynamic life cycle; resulting benefits can include workflow efficacy, decreased spending, streamlined inventory management, and increased patient safety. The RFID tags record data for each dose including lot number, NDC, and refrigeration date, which can help to centralize drug shortage and recall management, medication restocking, and inventory monitoring.